|
Soon after their announcement of polonium the Curies reported another radio-element, radium, on Boxing Day 1898. Litvinenko died after ten agonising days – as little as a microgram of polonium is fatal. In November 2006 someone added 210Po, probably as a chloride, to tea drunk in a London hotel by Alexander Litvinenko, an ex-KGB officer who had defected to the West. Recently a more sinister application was found for polonium. 
Irène and Frédéric Joliot-Curie used 210Po as an α-emitter for their own work on transmutation which earned them the 1935 Nobel prize in chemistry. Such uses are now rarer, but a Be- 210Po mixture is a useful neutron source. Polonium compounds were used as anti-static agents for gramophone records and for textile and photographic plate manufacture. Polonium exhibits early group 16-like behaviour in forming polonides, eg Na 2Po, which has the eight coordinate antifluorite structure. 
The commonest coordination numbers are six (octahedral or trigonal bipyramidal) and eight (cubic). The α and β forms of the element and its compounds emit a blue-white glow, and study of polonium chemistry is difficult because polonium is intensely toxic and the α-emission from 210Po attacks glassware and rapidly degrades its compounds.The electronic configuration of Po is 6p 4 and the principal oxidation states of its compounds are IV (predominant in solution), II and -II, with oxides, halides, sulfides etc of Po(IV) and Po(II). Group 16 elements show an increasing tendency to metallic behaviour with increasing atomic weight: oxygen and sulfur are non-metals, selenium and tellurium semi-conductors, but polonium is a soft, silvery-white metal with much higher electrical and thermal conductivities than tellurium. Most of the chemical work on polonium uses man-made 210Po as it is so rare in nature. There are 46 known radioactive isotopes of polonium 5 and the Curies had probably isolated 210Po, with a half-life ( t ½) of 138 days.īecause of its scarcity in nature 210Po is now normally made by neutron irradiation of bismuth in a nuclear reactor: Mendeleev had allocated a place for polonium in his 1889 periodic table as dvi -tellurium, predicting an atomic weight of 212 (modern value 209.98). 4 In a publication in 1902 on the nature of radioactivity they briefly cast doubt on the existence of polonium, but it was isolated later that year by Willy Marckwald. 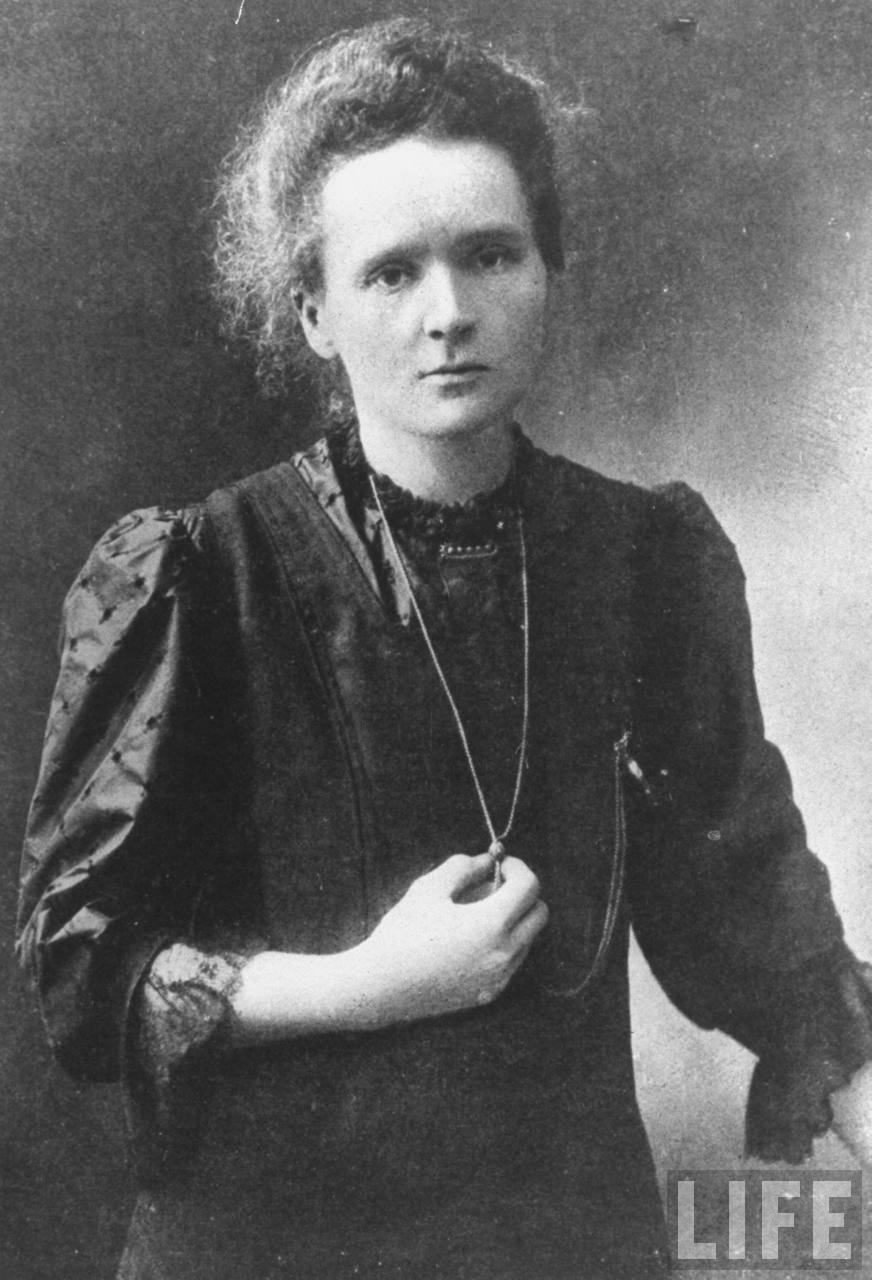
If the existence of this new element is confirmed, we propose to call it polonium from the country of origin of one of us.Ĭrucially, they also suggested that radioactivity was a property of the atom. In July the Curies published a paper, presented to the French Academy of Sciences by Becquerel, in which they coined the term radioactive. However, on sublimation minute traces of the radioactive sulfide condensed on a cooler part of the tube than the bismuth. Further manipulations showed that the sulfide of the new material co-precipitated with that of bismuth. On 14 April 1898 Marie dissolved 100g of pitchblende, UO 2, in HCl and passed H 2S into the solution the precipitated sulfides were still highly radioactive. Pitchblende, the source of uranium for the Curies 
3 At this point Pierre joined her research work, continuing until he was tragically killed in a road accident in 1906. She also showed that uranium ores were more active than pure uranium and so must contain other, stronger, radioactive constituents. Using one of Pierre's electrometers she showed, by studying a wide range of inorganic materials, that of the then known elements, only uranium and thorium were radioactive. After meeting Pierre Curie, a pioneer in piezoelectricity and magnetochemistry in 1894, the couple married in 1895.Īfter Henri Becquerel's discovery of radioactivity in 1896 1,2 Marie decided to work on 'Becquerel rays' for her doctorate. Advanced scientific studies were not then possible for Polish women so Marie followed her sister Bronia to Paris to study physics and chemistry at the Sorbonne in 1893–4. She showed an early interest in science and was encouraged in this by her parents who were both teachers. Marie 1,2 (1867–1934) was born in Warsaw as Maria Salomea Sklodowska.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
